Translations
| Code | Language | Translator | Run | |
|---|---|---|---|---|
 |
||||
Credits



 Written by Loo Kang Wee; Felix J. Garcia Clemente; Francisco Esquembre; Designed by David Loh
Written by Loo Kang Wee; Felix J. Garcia Clemente; Francisco Esquembre; Designed by David Loh
Overview:
This briefing document reviews the "A Level Chemical Bonding Dot and Cross Diagrams JavaScript Simulation Applet HTML5" available through Open Educational Resources / Open Source Physics @ Singapore, and the associated information provided on the "Chemical Bonding Dot and Cross Diagrams" page. The central theme is the provision of an interactive simulation tool designed to help students, particularly at the A Level (and a similar version for O Level), visualize and understand the formation of chemical bonds through the use of dot and cross diagrams.
Main Themes and Important Ideas/Facts:
- Interactive Learning Tool: The core of this resource is a JavaScript HTML5 simulation applet. The page explicitly states, "Embed this model in a webpage: ". This highlights the practical nature of the resource, allowing teachers to integrate it directly into their online learning platforms.
- Focus on Dot and Cross Diagrams: The applet is specifically designed to illustrate chemical bonding using dot and cross diagrams. This method visually represents the valence electrons of atoms and how they are shared or transferred during bond formation.
- Target Audience: The resource is explicitly mentioned as being for "A Level Chemical Bonding," with a note indicating that the "O level" version is similar except for the inclusion of the "sulfate(IV) ions, SO₃²⁻" question in the A Level version. This suggests the tool is tailored to specific curriculum levels.
- Illustrative Examples: The webpage provides a list of various molecules and ions for which the dot and cross diagrams are demonstrated. These include:
- Hydrogen molecule (H₂)
- Chlorine molecule (Cl₂)
- Oxygen molecule (O₂)
- Nitrogen molecule (N₂)
- Hydrogen Chloride molecule (HCl)
- Hydroxide ion (OH⁻)
- Cyanide ion (CN⁻)
- Water molecule (H₂O)
- Carbon Dioxide molecule (CO₂)
- Nitrite ion (NO₂⁻)
- Ammonia molecule (NH₃)
- Methane molecule (CH₄)
- Carbonate ion (CO₃²⁻)
- Hydrogen Peroxide (H₂O₂)
- Sulfate(IV) ions (SO₃²⁻) - specific to the A Level version.
- Learning Goals: The "Sample Learning Goals" section confirms the purpose of the simulation is to help students understand and visualize chemical bonding using dot and cross diagrams.
- Answers Provided: For each example molecule or ion listed, there is a brief "answer" provided, which describes the resulting electron arrangement in the outermost shells after bonding. For example, for the Hydrogen molecule, the answer is "add up to 2 on each H atom electron outermost with shared electrons = 2". This provides immediate feedback and clarifies the bonding in each case. Similarly, for the Chlorine molecule, the answer is "add up to 8 on each atom's' electron outermost shell with shared electrons = 2", demonstrating the octet rule.
- Credits and Development: The resource acknowledges the individuals involved in its creation: "Written by Loo Kang Wee; Felix J. Garcia Clemente; Francisco Esquembre; Designed by David Loh." This provides context and attribution for the work. The compilation using "EJS 6.0 (191124)" is also noted.
- Open Educational Resource: The platform is identified as "Open Educational Resources / Open Source Physics @ Singapore," and the content is licensed under a "Creative Commons Attribution-Share Alike 4.0 Singapore License." This indicates the resource is freely available for use and adaptation under certain conditions.
- Integration with Other Resources: The "For Teachers" section mentions a Google Slides presentation by David Loh that serves as a basis for the interactive simulation, providing a link for educators to access this related material.
- Part of a Larger Ecosystem: The webpage is embedded within a broader website offering various interactive resources for science and mathematics, as indicated by the breadcrumbs and the extensive list of "Other Resources" and "Popular Tags." This suggests that the dot and cross diagram applet is part of a larger effort to provide engaging and interactive educational content.
Key Quotes:
- (Regarding the embedding of the simulation): "Embed this model in a webpage: "
- (Sample Learning Goal): "A Level Chemical Bonding Dot and Cross Diagrams JavaScript Simulation Applet HTML5"
- (Answer for Hydrogen molecule): "answer is add up to 2 on each H atom electron outermost with shared electrons = 2"
- (Answer for Chlorine molecule): "answer is add up to 8 on each atom's' electron outermost shell with shared electrons = 2"
- (Licensing Information): "Contents are licensed Creative Commons Attribution-Share Alike 4.0 Singapore License ."
Conclusion:
The "A Level Chemical Bonding Dot and Cross Diagrams JavaScript Simulation Applet HTML5" is a valuable open educational resource for teaching and learning about chemical bonding. Its interactive nature, coupled with clear examples and provided answers, offers students a hands-on way to understand the formation of covalent and ionic bonds through dot and cross diagrams. The resource is well-attributed, openly licensed, and integrated within a larger collection of educational tools, making it a potentially useful asset for educators. The distinction between the A Level and O Level versions, particularly the inclusion of the sulfate(IV) ion in the former, highlights its curriculum-specific design.
Chemical Bonding: Dot and Cross Diagrams Study Guide
Quiz
- What is the purpose of using dot and cross diagrams in the study of chemical bonding?
- Describe the key difference in how valence electrons are represented in dot and cross diagrams for different atoms within a molecule.
- For a hydrogen molecule (H₂), how many electrons are shared between the two hydrogen atoms, and what is the resulting electron configuration around each atom?
- In a chlorine molecule (Cl₂), how many valence electrons does each chlorine atom contribute to the covalent bond, and what is the electron configuration around each chlorine atom after bonding?
- Explain why the oxygen molecule (O₂) requires a double covalent bond, illustrating with the number of shared electrons.
- How many covalent bonds are present in a nitrogen molecule (N₂), and how many electrons are shared in total?
- In the formation of a hydrogen chloride molecule (HCl), describe how the electrons are shared, and indicate if the sharing is equal or unequal.
- For the hydroxide ion (OH⁻), how does the overall negative charge affect the number of electrons shown in the dot and cross diagram?
- In the carbonate ion (CO₃²⁻), describe the arrangement of electrons around the carbon and oxygen atoms, including any lone pairs and shared electrons.
- How do dot and cross diagrams help to visualize the achievement of a stable electron configuration (usually an octet or duet) in molecules and ions?
Quiz Answer Key
- Dot and cross diagrams visually represent the valence electrons of atoms within a molecule or ion and how these electrons are shared or transferred during chemical bonding. This helps in understanding the formation of covalent and ionic bonds and the resulting electron configurations.
- Typically, dots represent the valence electrons originating from one type of atom, while crosses represent the valence electrons from the other type of atom involved in the bond. This distinction allows visualization of electron sharing or transfer.
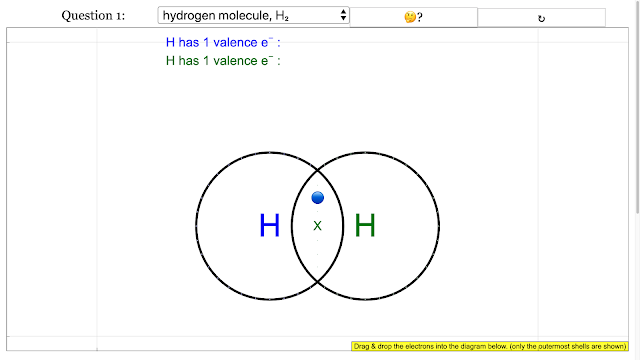
- In a hydrogen molecule, two electrons are shared, forming a single covalent bond. This results in each hydrogen atom having two electrons in its outermost shell, achieving a stable duet configuration similar to helium.
- In a chlorine molecule, each chlorine atom contributes one valence electron to form a single covalent bond, resulting in a shared pair of electrons. Each chlorine atom then has eight electrons in its outermost shell, achieving a stable octet configuration.
- The oxygen molecule requires a double covalent bond because each oxygen atom has six valence electrons and needs two more to achieve a stable octet. They share four electrons (two pairs), forming two covalent bonds between them.
- A nitrogen molecule (N₂) has a triple covalent bond, meaning three pairs of electrons are shared between the two nitrogen atoms. This results in a total of six shared electrons and an octet of electrons around each nitrogen atom.
- In HCl, hydrogen and chlorine share a pair of electrons, forming a single covalent bond. The sharing is unequal because chlorine is more electronegative than hydrogen, pulling the shared electrons closer to the chlorine atom.
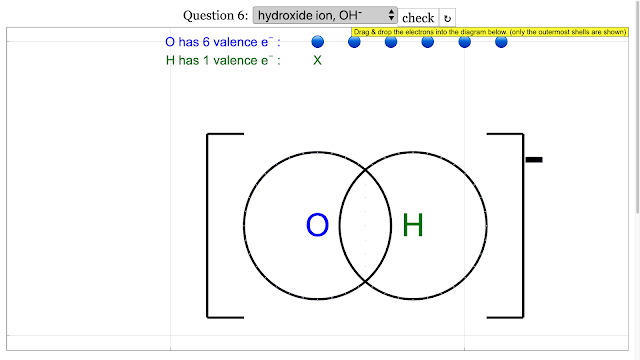
- For the hydroxide ion (OH⁻), the overall negative charge indicates the presence of an extra electron in the ion. This additional electron is shown in the dot and cross diagram, usually associated with the oxygen atom, contributing to its stable octet.
- In the carbonate ion (CO₃²⁻), the carbon atom typically forms double and single bonds with oxygen atoms, sharing a total of four electrons. Each oxygen atom achieves an octet, with some oxygen atoms also possessing lone pairs and contributing to the overall negative charge through extra electrons.
- Dot and cross diagrams illustrate how atoms achieve a stable electron configuration (octet or duet) by showing the redistribution of valence electrons through the sharing in covalent bonds or the transfer in ionic bonds, satisfying the octet rule for most atoms.
Essay Format Questions
- Discuss the strengths and limitations of using dot and cross diagrams to represent chemical bonding. In what situations are these diagrams most helpful, and when might other models of bonding be more appropriate?
- Explain the octet rule and how it is reflected in dot and cross diagrams for various molecules and polyatomic ions presented in the source material. Are there any exceptions to the octet rule illustrated in these examples?
- Compare and contrast the formation of covalent bonds in simple diatomic molecules (like H₂, Cl₂) with covalent bonding in polyatomic ions (like OH⁻, CO₃²⁻) as depicted by dot and cross diagrams. Highlight the role of formal charges and overall charge in the diagrams of polyatomic ions.
- Analyze the dot and cross diagrams for molecules with single, double, and triple bonds (e.g., Cl₂, O₂, N₂). How does the number of shared electron pairs relate to the stability and properties of these molecules?
- Based on the provided list of molecules and ions, describe the general steps involved in drawing a dot and cross diagram for a given chemical species. What information about the constituent atoms is essential before constructing these diagrams?
Glossary of Key Terms
- Valence Electrons: The electrons in the outermost shell of an atom, which are primarily involved in chemical bonding.
- Dot and Cross Diagram (Lewis Structure): A visual representation of the valence electrons of atoms in a molecule or ion, where dots and crosses are used to distinguish electrons from different atoms and show how they are shared or transferred in chemical bonds.
- Covalent Bond: A chemical bond that involves the sharing of one or more pairs of electrons between atoms.
- Single Bond: A covalent bond in which one pair of electrons is shared between two atoms.
- Double Bond: A covalent bond in which two pairs of electrons (four electrons) are shared between two atoms.
- Triple Bond: A covalent bond in which three pairs of electrons (six electrons) are shared between two atoms.
- Lone Pair (Non-bonding Electrons): A pair of valence electrons that are not involved in forming covalent bonds and are associated with a single atom.
- Octet Rule: The tendency of atoms to prefer to have eight electrons in their valence shell to achieve greater stability, similar to the electron configuration of noble gases.
- Duet Rule: The tendency of some small atoms like hydrogen and lithium to achieve a stable configuration with two valence electrons.
- Polyatomic Ion: An ion composed of two or more atoms covalently bonded together and carrying an overall electric charge.
- Chemical Formula: A symbolic representation of a chemical compound that shows the types of atoms and their ratios in the compound.
- Electron Configuration: The arrangement of electrons within the energy levels and sublevels of an atom.
- Electronegativity: A measure of the tendency of an atom to attract a bonding pair of electrons. Unequal sharing of electrons in a covalent bond occurs when there is a difference in electronegativity between the bonded atoms.
Sample Learning Goals
This simulation is the same as the O level one except for the last question "sulfate(IV) ions, SO₃²⁻"
- A Level Chemical Bonding Dot and Cross Diagrams JavaScript Simulation Applet HTML5
- O level Chemical Bonding Dot and Cross Diagrams JavaScript Simulation Applet HTML5
For Teachers
From Google slides (From David) to interactive https://docs.google.com/presentation/d/1fwutLc-jPc1fUyrxJsps3Fhg6_J9y8AEA4pHJ68gdBw/edit?ts=5dd2086a#slide=id.g5292a6c619_0_96
Chemical Bonding Dot and Cross Diagrams
Polyatomic ions dot and cross diagram
|
Chemical Bonding Dot and Cross Diagrams for Hydrogen molecule |
|
Chemical Bonding Dot and Cross Diagrams for Hydrogen molecule answer is add up to 2 on each H atom electron outermost with shared electrons = 2 |
|
Chemical Bonding Dot and Cross Diagrams for Chlorine molecule |
|
Chemical Bonding Dot and Cross Diagrams for Chlorine molecule answer is add up to 8 on each atom's' electron outermost shell with shared electrons = 2 |
|
Chemical Bonding Dot and Cross Diagrams for Oxygen molecule |
|
Chemical Bonding Dot and Cross Diagrams for Oxygen molecule answer is is add up to 8 on each atom's' electron outermost shell with shared electrons = 4 |
|
Chemical Bonding Dot and Cross Diagrams for Nitrogen molecule |
|
Chemical Bonding Dot and Cross Diagrams for Nitrogen molecule answer is is add up to 8 on each atom's' electron outermost shell with shared electrons = 6 |
|
Chemical Bonding Dot and Cross Diagrams for Hydrogen Chloride molecule |
Frequently Asked Questions: Dot and Cross Diagrams for Chemical Bonding
- What is the purpose of a dot and cross diagram in chemistry? A dot and cross diagram is a visual representation used to illustrate the transfer or sharing of valence electrons (outermost shell electrons) between atoms during the formation of chemical bonds. Dots typically represent the valence electrons of one type of atom, while crosses represent the valence electrons of the other atom(s) involved in the bonding. This helps to visualize how atoms achieve a stable electron configuration, usually resembling that of a noble gas (typically 8 valence electrons, or 2 for hydrogen and lithium).
- What types of chemical bonds can be represented using dot and cross diagrams? Dot and cross diagrams are primarily used to represent covalent bonds (where electrons are shared between atoms) and ionic bonds (where electrons are transferred from one atom to another, resulting in the formation of ions with opposite charges that attract each other). They can also be used for polyatomic ions, showing both the covalent bonds within the ion and the overall charge due to a gain or loss of electrons.
- How do you draw a dot and cross diagram for a simple covalent molecule like hydrogen (H₂)? For a hydrogen molecule, each hydrogen atom has one valence electron. In the dot and cross diagram, one hydrogen atom's electron is represented by a dot, and the other hydrogen atom's electron is represented by a cross. These two electrons are shown to be shared in an overlapping region between the two hydrogen atoms, indicating a single covalent bond. Each hydrogen atom effectively has 2 electrons in its outer shell through this sharing.
- How does the dot and cross diagram for oxygen (O₂) differ from that of hydrogen (H₂)? An oxygen atom has six valence electrons. In an oxygen molecule (O₂), two oxygen atoms share two pairs of electrons to form a double covalent bond. The dot and cross diagram would show each oxygen atom with its six original valence electrons (represented by dots for one oxygen and crosses for the other), with two pairs of electrons (two dots and two crosses) being shared in the overlapping region between the atoms. This sharing allows each oxygen atom to achieve a stable octet (8 valence electrons).
- How are ionic compounds, like sodium chloride (NaCl), represented with dot and cross diagrams? In ionic bonding, there is a transfer of electrons rather than sharing. Sodium (Na) has one valence electron, and chlorine (Cl) has seven. In the formation of sodium chloride, sodium loses its one valence electron to become a positively charged sodium ion (Na⁺) with a stable electron configuration. Chlorine gains this electron to become a negatively charged chloride ion (Cl⁻), also achieving a stable octet. The dot and cross diagram would show sodium with no valence electrons around it (and a positive charge indicated), and chlorine with eight valence electrons around it (including the transferred electron, and a negative charge indicated). The transferred electron would be represented by the symbol (dot or cross) of the original atom.
- How do you represent polyatomic ions, such as the hydroxide ion (OH⁻), using dot and cross diagrams? For a polyatomic ion like hydroxide (OH⁻), you first represent the covalent bond between oxygen and hydrogen, showing the sharing of electrons. Oxygen has six valence electrons and hydrogen has one. They share one pair of electrons. The negative charge on the hydroxide ion indicates that it has gained an additional electron. This extra electron would be shown on the oxygen atom (usually with a different symbol than its original valence electrons or the shared electrons), and the entire structure would be enclosed in brackets with a "-1" charge indicated outside. The oxygen atom should have a total of eight electrons around it (including shared and its own/gained), and hydrogen should have two (shared).
- What is the significance of achieving a noble gas electron configuration in dot and cross diagrams? The driving force behind chemical bonding is the tendency of atoms to achieve a stable electron configuration, which is typically the same as that of a noble gas. Noble gases have a full outer shell of electrons (usually eight, or two for helium), making them very stable and unreactive. In dot and cross diagrams, the goal is to show how atoms, through sharing or transferring electrons, can achieve this stable arrangement, resulting in the formation of molecules or ions.
- Can the provided simulation tool help in understanding dot and cross diagrams? Yes, the description explicitly mentions a "A Level Chemical Bonding Dot and Cross Diagrams JavaScript Simulation Applet HTML5". This interactive tool allows users to visualize and manipulate dot and cross diagrams for various molecules and ions, including more complex examples like sulfate(IV) ions (SO₃²⁻), hydrogen peroxide (H₂O₂), and carbonate ions (CO₃²⁻). It can provide a dynamic and hands-on way to learn and understand the arrangement of valence electrons in chemical bonds.
|
Chemical Bonding Dot and Cross Diagrams for Hydrogen Chloride molecule answer is is add up to 8 on Cl atom and 2 for H atom and electron outermost shell with shared electrons = 2 |
|
Chemical Bonding Dot and Cross Diagrams for Hydroxide ion |
|
Chemical Bonding Dot and Cross Diagrams for Hydroxide ion answer is is add up to 8 on O atom and 2 for H atom and electron outermost shell with shared electrons = 2 |
|
Chemical Bonding Dot and Cross Diagrams for Cyanide ion |
|
Chemical Bonding Dot and Cross Diagrams for Cyanide ion answer is is add up to 8 on each atoms and electron outermost shell with shared electrons = 6 |
|
Chemical Bonding Dot and Cross Diagrams for Water molecule |
|
Chemical Bonding Dot and Cross Diagrams for Water molecule answer is is add up to 8 on O atom and 2 on H atoms and electron outermost shell with shared electrons = 2,2 respectively |
|
Chemical Bonding Dot and Cross Diagrams for Carbon Dioxide molecule |
|
Chemical Bonding Dot and Cross Diagrams for Carbon Dioxide molecule answer is is add up to 8 on each atom and electron outermost shell with shared electrons = 4,4 respectively |
|
Chemical Bonding Dot and Cross Diagrams for Nitrite ion |
|
Chemical Bonding Dot and Cross Diagrams for Nitrite ion answer is is add up to 8 on each atom and electron outermost shell with shared electrons = 2,4 respectively with the foreign electron on the O atom with the shared electrons=2 |
|
Chemical Bonding Dot and Cross Diagrams for Nitrite ion answer is is add up to 8 on each atom and electron outermost shell with shared electrons = 2,4 respectively with the foreign electron on the O atom with the shared electrons=2 |
|
Chemical Bonding Dot and Cross Diagrams for Ammonia molecule |
|
Chemical Bonding Dot and Cross Diagrams for Ammonia molecule answer is is add up to 8 on N atom and 2 on H atom and the electron outermost shell with shared electrons = 2,2,2 respectively |
|
Chemical Bonding Dot and Cross Diagrams for Methane molecule |
|
Chemical Bonding Dot and Cross Diagrams for Methane molecule answer is is add up to 8 on C atom and 2 on H atom and the electron outermost shell with shared electrons = 2,2,2, respectively |
|
Chemical Bonding Dot and Cross Diagrams for Carbonate ion |
|
Chemical Bonding Dot and Cross Diagrams for Carbonate ion answer is is add up to 8 on each atom and the electron outermost shell with shared electrons = 4,2,2, respectively. The two O atoms with 2 shared electrons has a foreign electron in it's personal shell. |
 |
|
Chemical Bonding Dot and Cross Diagrams for Hydrogen Peroxide |
Video
[text]
Version:
- https://weelookang.blogspot.com/2019/12/chemical-bonding-dot-and-cross-diagrams.html
- https://sites.google.com/a/mgs.sch.edu.sg/about-me-david-loh/
Other Resources
[text]
Frequently Asked Questions: Dot and Cross Diagrams for Chemical Bonding
- What is the purpose of a dot and cross diagram in chemistry? A dot and cross diagram is a visual representation used to illustrate the transfer or sharing of valence electrons (outermost shell electrons) between atoms during the formation of chemical bonds. Dots typically represent the valence electrons of one type of atom, while crosses represent the valence electrons of the other atom(s) involved in the bonding. This helps to visualize how atoms achieve a stable electron configuration, usually resembling that of a noble gas (typically 8 valence electrons, or 2 for hydrogen and lithium).
- What types of chemical bonds can be represented using dot and cross diagrams? Dot and cross diagrams are primarily used to represent covalent bonds (where electrons are shared between atoms) and ionic bonds (where electrons are transferred from one atom to another, resulting in the formation of ions with opposite charges that attract each other). They can also be used for polyatomic ions, showing both the covalent bonds within the ion and the overall charge due to a gain or loss of electrons.
- How do you draw a dot and cross diagram for a simple covalent molecule like hydrogen (H₂)? For a hydrogen molecule, each hydrogen atom has one valence electron. In the dot and cross diagram, one hydrogen atom's electron is represented by a dot, and the other hydrogen atom's electron is represented by a cross. These two electrons are shown to be shared in an overlapping region between the two hydrogen atoms, indicating a single covalent bond. Each hydrogen atom effectively has 2 electrons in its outer shell through this sharing.
- How does the dot and cross diagram for oxygen (O₂) differ from that of hydrogen (H₂)? An oxygen atom has six valence electrons. In an oxygen molecule (O₂), two oxygen atoms share two pairs of electrons to form a double covalent bond. The dot and cross diagram would show each oxygen atom with its six original valence electrons (represented by dots for one oxygen and crosses for the other), with two pairs of electrons (two dots and two crosses) being shared in the overlapping region between the atoms. This sharing allows each oxygen atom to achieve a stable octet (8 valence electrons).
- How are ionic compounds, like sodium chloride (NaCl), represented with dot and cross diagrams? In ionic bonding, there is a transfer of electrons rather than sharing. Sodium (Na) has one valence electron, and chlorine (Cl) has seven. In the formation of sodium chloride, sodium loses its one valence electron to become a positively charged sodium ion (Na⁺) with a stable electron configuration. Chlorine gains this electron to become a negatively charged chloride ion (Cl⁻), also achieving a stable octet. The dot and cross diagram would show sodium with no valence electrons around it (and a positive charge indicated), and chlorine with eight valence electrons around it (including the transferred electron, and a negative charge indicated). The transferred electron would be represented by the symbol (dot or cross) of the original atom.
- How do you represent polyatomic ions, such as the hydroxide ion (OH⁻), using dot and cross diagrams? For a polyatomic ion like hydroxide (OH⁻), you first represent the covalent bond between oxygen and hydrogen, showing the sharing of electrons. Oxygen has six valence electrons and hydrogen has one. They share one pair of electrons. The negative charge on the hydroxide ion indicates that it has gained an additional electron. This extra electron would be shown on the oxygen atom (usually with a different symbol than its original valence electrons or the shared electrons), and the entire structure would be enclosed in brackets with a "-1" charge indicated outside. The oxygen atom should have a total of eight electrons around it (including shared and its own/gained), and hydrogen should have two (shared).
- What is the significance of achieving a noble gas electron configuration in dot and cross diagrams? The driving force behind chemical bonding is the tendency of atoms to achieve a stable electron configuration, which is typically the same as that of a noble gas. Noble gases have a full outer shell of electrons (usually eight, or two for helium), making them very stable and unreactive. In dot and cross diagrams, the goal is to show how atoms, through sharing or transferring electrons, can achieve this stable arrangement, resulting in the formation of molecules or ions.
- Can the provided simulation tool help in understanding dot and cross diagrams? Yes, the description explicitly mentions a "A Level Chemical Bonding Dot and Cross Diagrams JavaScript Simulation Applet HTML5". This interactive tool allows users to visualize and manipulate dot and cross diagrams for various molecules and ions, including more complex examples like sulfate(IV) ions (SO₃²⁻), hydrogen peroxide (H₂O₂), and carbonate ions (CO₃²⁻). It can provide a dynamic and hands-on way to learn and understand the arrangement of valence electrons in chemical bonds.
- Details
- Written by David Loh
- Parent Category: Chemistry
- Category: 03 Chemistry of Reactions
- Hits: 9900


.png
)