About
Topics
- Chemical Equations
- Conservation of Mass
Description
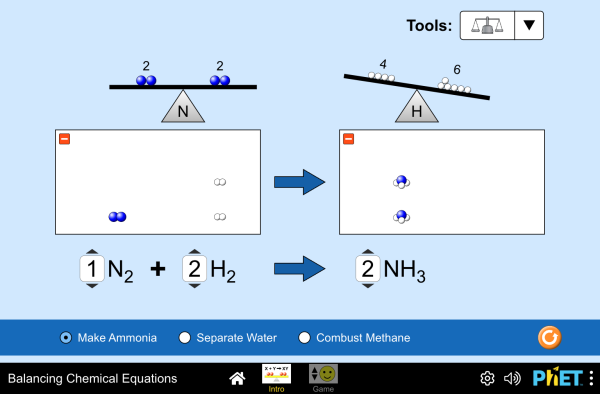
How do you know if a chemical equation is balanced? What can you change to balance an equation? Play a game to test your ideas!
Sample Learning Goals
- Balance a chemical equation.
- Recognize that the number of atoms of each element is conserved in a chemical reaction.
- Describe the difference between coefficients and subscripts in a chemical equation.
- Translate from symbolic to molecular representations of matter.
For Teachers
[SIMU_TEACHER]
Software Requirements
| Windows 7+ | Mac OS 10.7+ | iPad and iPad Mini with iOS | Chromebook with Chrome OS |
|---|---|---|---|
|
Internet Explorer 10+
latest versions of Chrome and Firefox
|
Safari 6.1 and up
latest versions of Chrome and Firefox
|
latest version of Safari | latest version of Chrome |
Credits
| Design Team | Third-party Libraries | Thanks To |
|---|---|---|
|
|